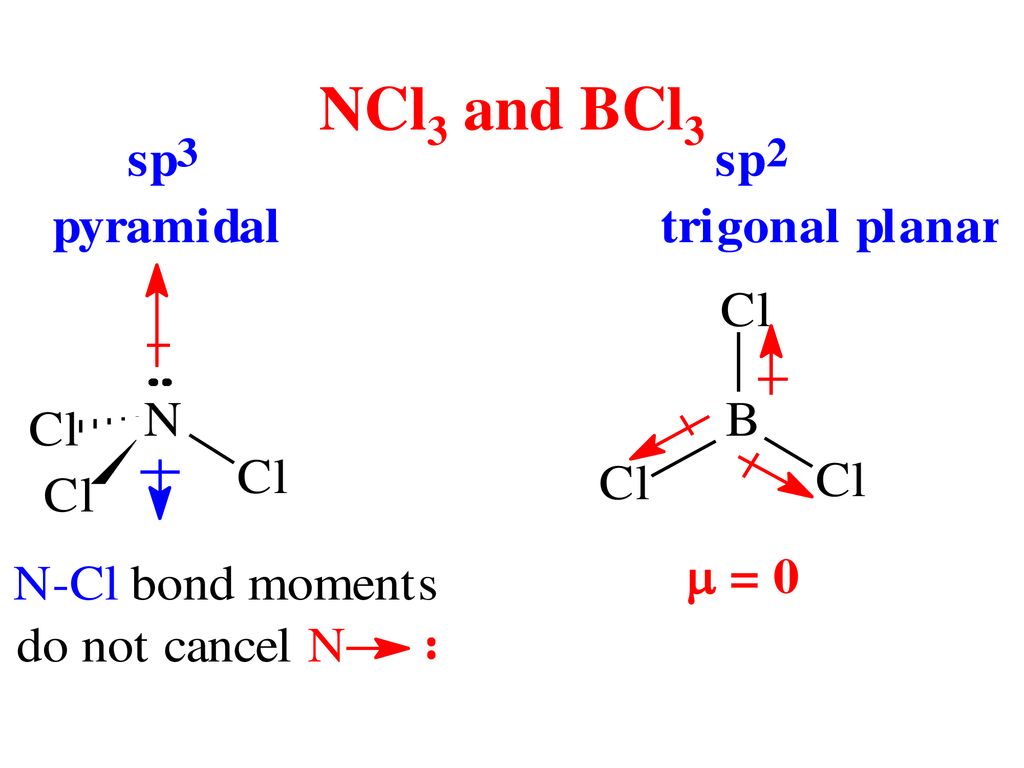
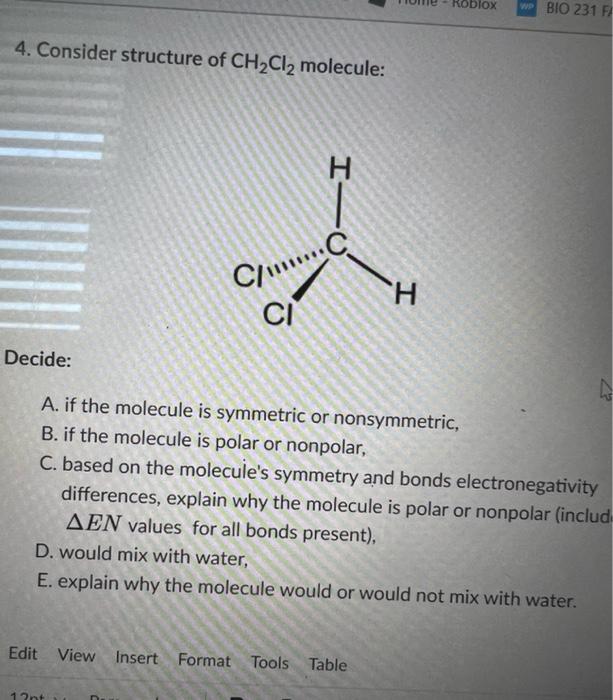
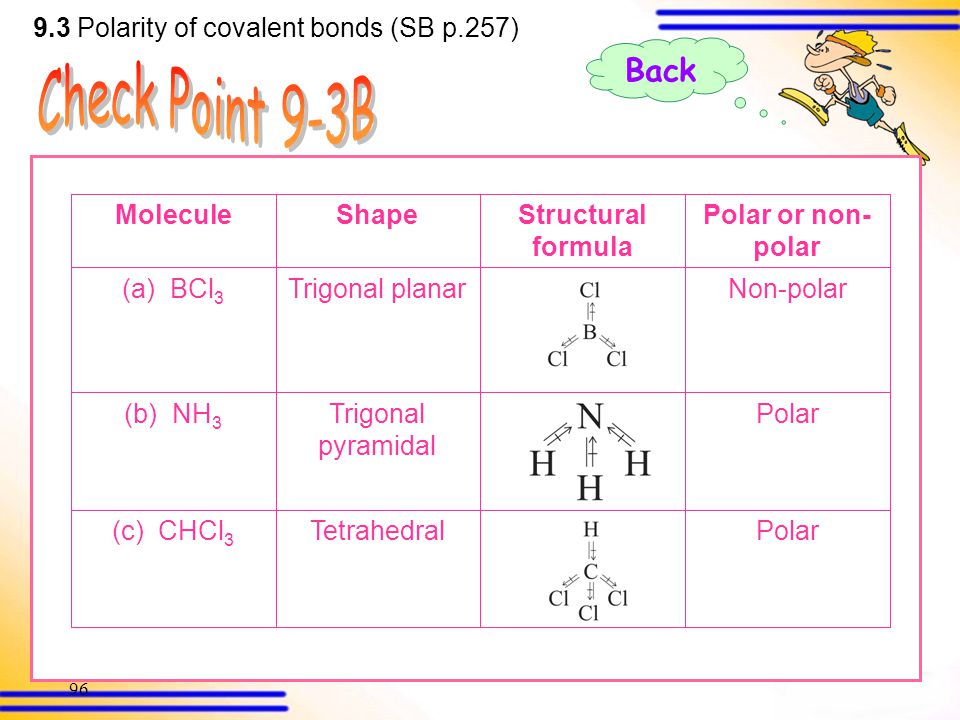
Which of the following molecules are nonpolar? (Select all that apply.) a. BCl3 b. O2 c. CH2O d. O3 | Homework.Study.com

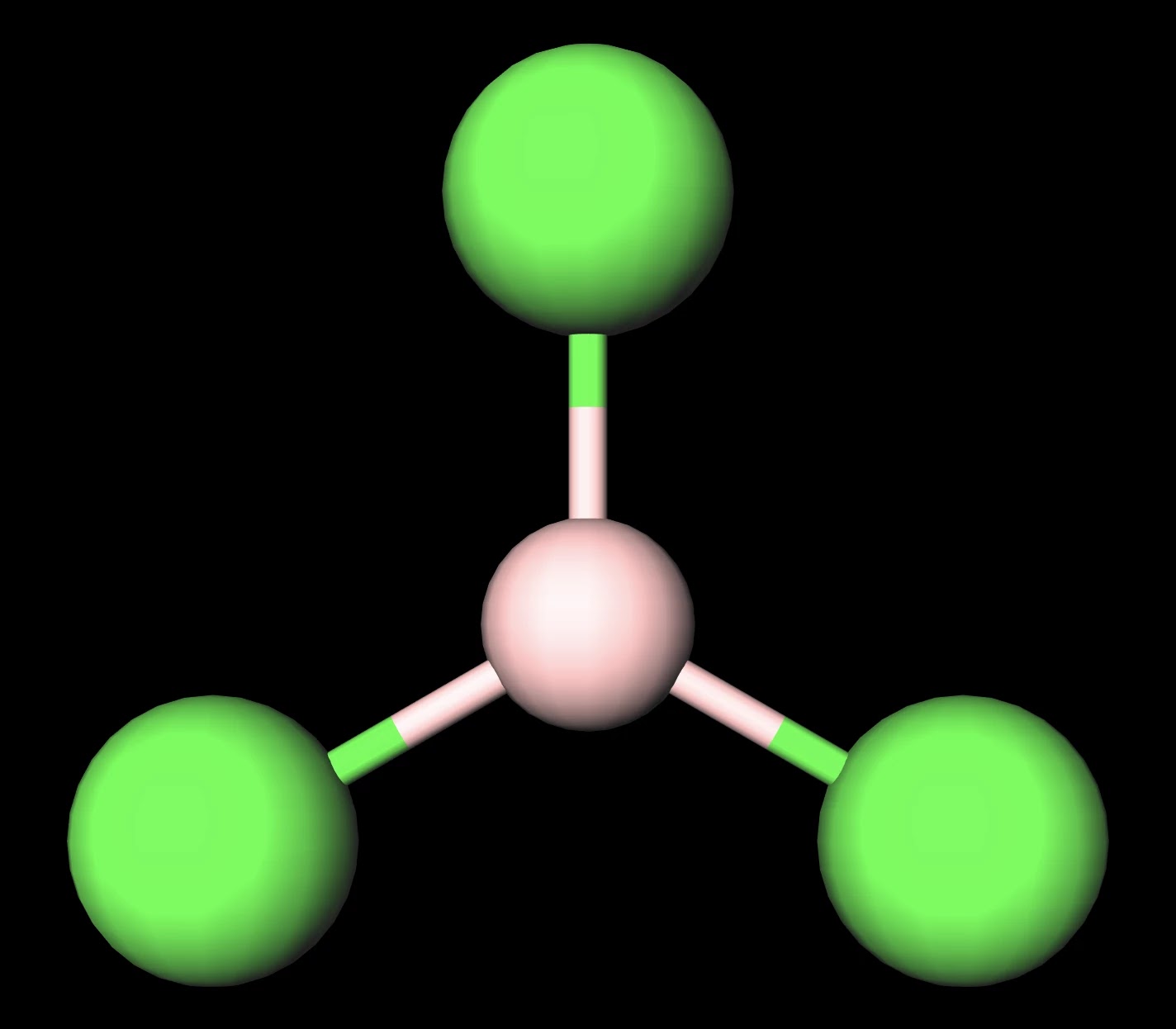
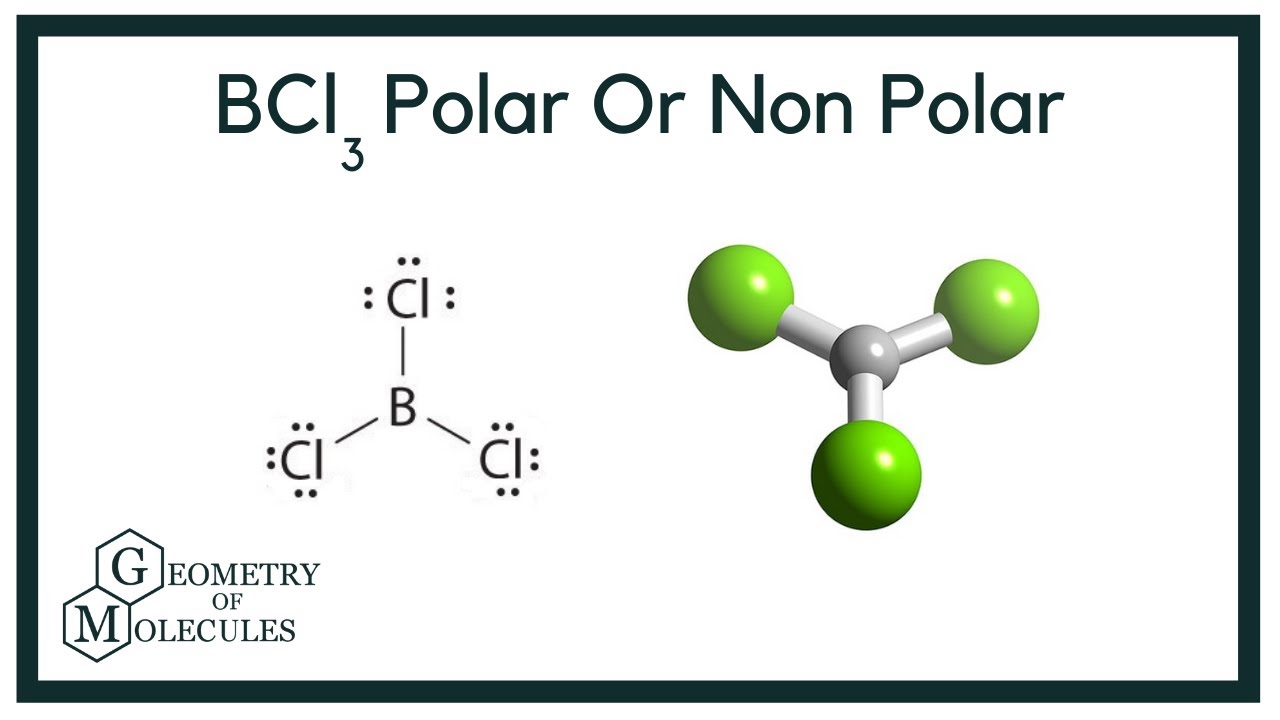
BCl3 lewis structure, molecular geometry, polar or nonpolar, hybridization, Bond angle | Molecular geometry, Molecular, Vsepr theory

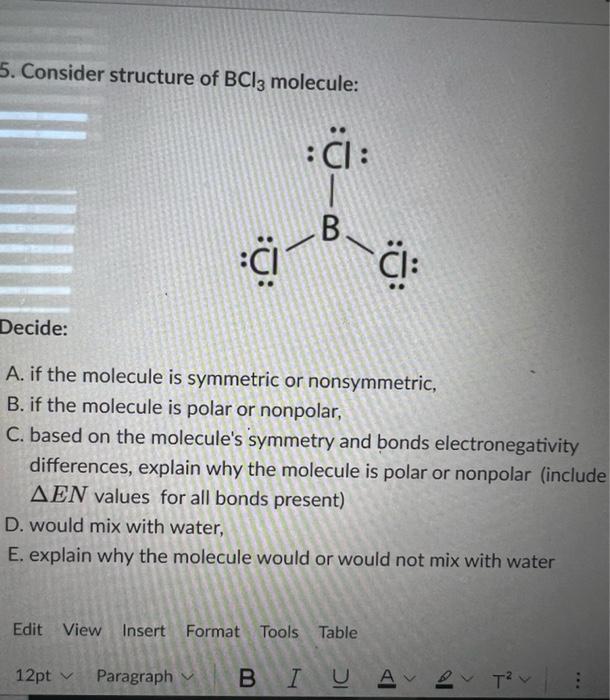
Draw the Lewis structure for BCl3. Determine its electron geometry, the number of non-bonding domains on the central atom, and the polarity of the molecule. | Homework.Study.com

BCl3 Lewis Structure (Boron Trichloride) | BCl3 Lewis Structure (Boron Trichloride) Welcome back to our channel and in today's video we will help you determine the Lewis Structure of Boron... | By